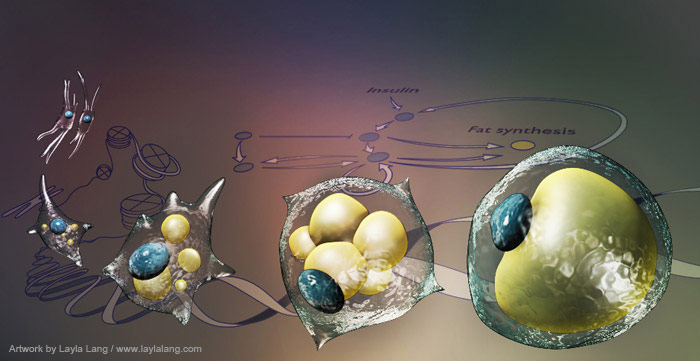
Precise decision-making is critical in differentiation where decisions often result in an irreversible switch of cell-fate that, if incorrectly made, can have consequences for human health. We are working to understand how to control cell differentiation decisions using adipogenesis as model system.
Adipogenesis as a Model System
 Figure 1: Ten percent of every adult human’s fat cells are replaced each year (Spalding et al, Nature 2010). What molecular mechanisms sense and set this constant rate of cell differentiation? Can we control this rate to treat obesity and metabolic disease?
Figure 1: Ten percent of every adult human’s fat cells are replaced each year (Spalding et al, Nature 2010). What molecular mechanisms sense and set this constant rate of cell differentiation? Can we control this rate to treat obesity and metabolic disease?Differentiated human cells are continuously replaced due to age-related damage. Adipocytes (fat cells) make up 25% of our body mass and are replaced at a rate of ~10% per year. We are using adipocytes as a model system to understand the regulatory circuit of cell differentiation, not only because adipocyte-related diseases are on the rise, but also because adipocytes can be readily studied using powerful live-cell microscopy and quantitative mass spectrometry approaches. A main goal of our lab is to apply these approaches to answer the fundamental question of how cells build regulatory circuits to make reliable and irreversible cell fate decisions.
We have identified a system that combines multiple positive transcriptional and signaling feedbacks to make irreversible differentiation decisions at the single cell level. At the same time, we discovered that cells use stochastic expression noise in these feedbacks loops to allow organisms to regulate the fraction of precursor cells that differentiate over time to control long term body weight. Many fundamental questions remain to be addressed about the molecular steps in this fundamental process which integrate different signaling pathways, transcription factors and chromatin remodeling proteins.
Translating our Basic Science Discoveries into Treatments for Insulin-resistance, Diabetes, Obesity, and Fat-related Cancers
Currently more than 25% of American adults have insulin resistance, a condition which results in a substantially increased risk of developing diabetes and cardiovascular disease, the leading cause of death in the US. Of particular concern, childhood obesity is becoming more prevalent in the US, and an estimated 60% of these obese children will go on to develop insulin resistance. However, currently there are no treatments available for pediatric patients, and even for adults, the few existing treatments are often not effective. New drugs and approaches are needed to treat insulin resistance.
We are in a unique position to apply our in vitro findings of how differentiation is controlled into studies of mouse models of adipogenesis related diseases and also by investigating adipocyte tissue from human patients. We already showed that we can use our quantitative mass spectrometry approach to determine differences in key regulatory proteins in samples from mouse models and are currently extending this approach to human patient samples. Our goal is to use our new strategies to classify patients into different insulin resistance subtypes that may be responsive to patient specific effective drug treatments. This will allow us to translate the discoveries from our in vitro studies into improved therapies for insulin resistance, diabetes, cardiovascular disease, and the many fat-related cancers.
Relevant Publications
Specific to adipogenesis/insulin resistance/lipid storage:
Park BO, Ahrends R, Teruel MN. (2012). Consecutive positive feedback loops create a bistable switch that controls preadipocyte to adipocyte conversion. Cell Reports Oct 25; 2(4): 976-90. Epub 2012 Oct. 11. PMID: 23063366. [Pubmed Link]
Ahrends R, Ota A, Kovary KM, Kudo T, Park BO, Teruel MN. (2014). Controlling low rates of cell differentiation through noise and ultra-high feedback. Science Jun 20; 344:1384-9. PMID: 24948735. Awarded an Editors’ Choice rating by the Science magazine signaling editors.[Pubmed Link]
Khor VK, Ahrends R, Shen W, Cortez Y, Teruel MN, Salman A, and Kraemer FB. (2014). The proteome of cholesteryl-ester-enriched versus triacylglycerol-enriched lipid droplets. Plos One. Aug 11; 9(8):e105047. PMID: 25111084.[Pubmed Link]
Ota A, Kovary KM, Wu OH, Ahrends R, Costa MJ, Shen W, Feldman BJ, Kraemer FB, Teruel MN. (2015). Using SRM mass spectrometry to profile nuclear protein abundance differences between adipose tissue depots of insulin resistant mice. Journal of Lipid Research 56(5):1068-78. PMID: 25840986.[Pubmed Link]
Understanding, quantifying, and using cell-to-cell variability and feedback regulation in cells to regulate cell fate:
Kovary KM, Taylor B, Zhao ML, Teruel MN. (2018). Expression variation and covariation impair analog and enable binary signaling control. Molecular Systems Biology May 14;14(5):e7997. DOI: 10.15252/msb.20177997. PubMed PMID: 29759982; PubMed Central PMCID: PMC5951153.[Pubmed Link]
Shi Z, Fujii K, Kovary KM, Genuth NR, Röst HL, Teruel MN, Barna M. (2017). Heterogeneous Ribosomes Preferentially Translate Distinct Subpools of mRNAs Genome-wide. Molecular Cell Jul 6;67(1):71-83.e7. Epub 2017 Jun 15. PubMed PMID: 28625553.[Pubmed Link]
Abell E*, Ahrends R*, Bandara S, Park BO, Teruel MN. (2011). Parallel adaptive feedback enhances reliability of the Ca2+ signaling system. Proc Natl Acad Sci U S A. Aug 30; 108(35): 14485-90. Epub 2011 Aug 15. *equal contribution. Awarded a “Must Read” and “Exceptional” rating by the Faculty of 1000.[Pubmed Link]